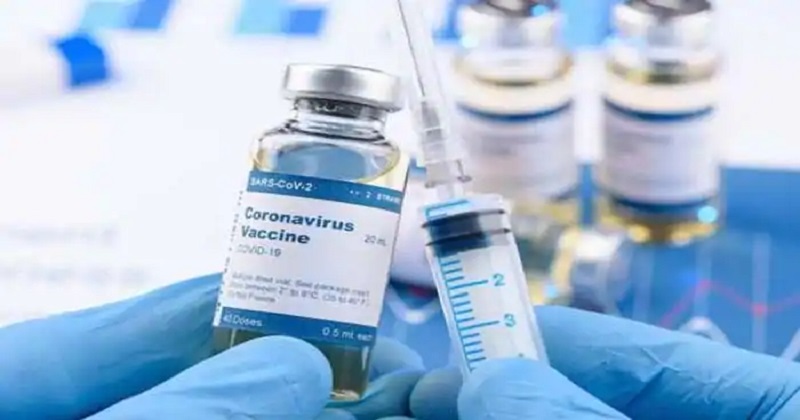
The UAE’s Ministry of Health and Prevention, Mohap, has informed the official registration of the Beijing Institute of Biological Product’s inactivated Covid-19 vaccine is a significant action towards fighting the global pandemic. The registration of this vaccine is a determination in reply to the application from Sinopharm CNBG. The notification is a significant vote of faith by the UAE’s health authorities in the safety and effectiveness of this vaccine.
Mohap in partnership with the Department of Health Abu Dhabi (DOH) has examined Sinopharm CNBG’s temporary examination of the phase III attempts, which depicts the Beijing Institute of Biological Product’s inactivated vaccine to have 86 percent effectiveness against Covid-19 illness. The examination also shows the vaccine to have a 99 percent seroconversion ratio of balancing antibody and 100 percent efficacy in controlling mild and severe cases of the disease. Also, the study indicates no severe safety problems.
In July that the Phase 3 clinical trials of the vaccine started in Abu Dhabi. Volunteers were allocated the vaccine or placebo. The rate of the test vaccines to the placebo is 2:1. Volunteers were inoculated with either Vaccine 1 (WIBP), Vaccine 2 (BIBP), or placebo. Two doses of vaccine were issued in a gap of 21 days. More than 31,000 volunteers from more than 120 nationalities took the medicine in just six weeks.
The trials are being handled by Abu-Dhabi based G42 Healthcare in partnership with the ministry, the Department of Health Abu Dhabi (DoH), and Abu Dhabi Health Services Company (Seha). This vaccine was vested Emergency Use Authorisation (EUA) in September by Mohap to save frontline workers most at threat of Covid-19. The UAE is operating Post Authorisation Safety Study (PASS) and Post Authorisation Efficacy Study (PAES) of its EUA program. These continuing studies show comparable safety and effectiveness profiles as the temporary analysis.
Read more; ‘Breaking news’ ; Maoist leader arrested from a hospital in Kerala !!!
The #4Humanity phase III trials have included 31,000 volunteers across 125 nationalities in the UAE alone. The EUA vaccination program has effectively safeguarded the UAE frontline workers. Mohap’s official registration of the Beijing Institute of Biological Product’s inactivated Covid-19 vaccine guides the path to completely saving the population and responsibly unlocking the economy. Individually, the UAE has established the volunteer program for Phase 3 clinical tests of the Russian Sputnik V Covid-19 vaccine. Now, only 500 volunteers from Abu Dhabi are asked to partake in the trials.

Post Your Comments