Many countries have already started screening tests when the Indian government says it will not change its vaccination agreements until mixed vaccines are scientifically supported. If the tests are successful, you will not need to delay your second dose just because a specific vaccine is not available. With early test results advising that a particular combination is safer and more apt to work, some countries have already selected such standards of ‘mixing and matching’ in emergencies.
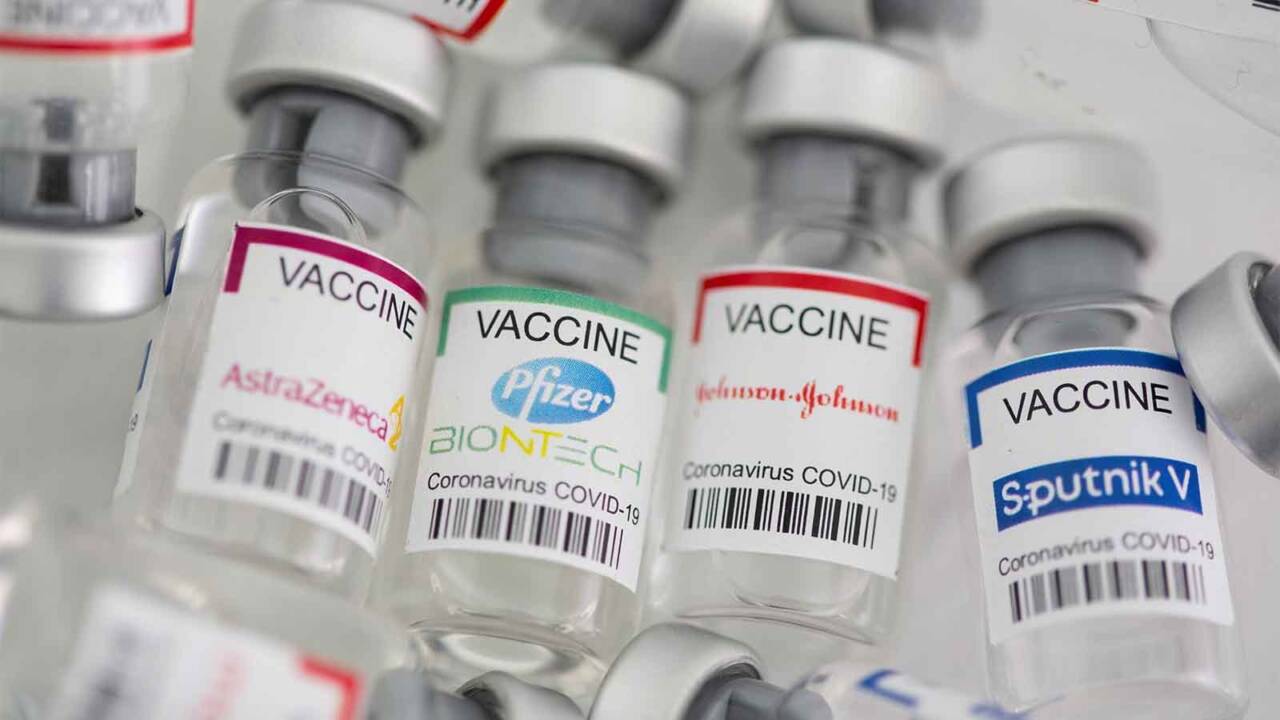
Except for Johnson & Johnson vaccine, all approved vaccines require two doses. Normally, the first dose triggers the immune system to recognize the virus and the second to improve its exposure and response.
So as to combine vaccines, consider the first dose of an antiretroviral drug-like Oxford-AstraZeneca’s Covishield, and the second dose of mRNA jab like Pfizer-BioNTech, called ‘heterologous prime-boost’ and can train the immune system to detect the virus in more than one way.
Regarding Sputnik V of Russia, it is a ‘combination’ vaccine by design. The first dose uses a harmless cold adenovirus (Ad26) to deliver genetic instructions for cells to produce the coronavirus’s spike protein. The second is to do it with a different adenovirus (Ad5), to avoid an attack of the immune system that now sees Ad26. This machine works – Sputnik V has a 91.6% performance rate, similar to the Pfizer and Moderna mRNA vaccines, even though it is an HIV vector-like Covishield.
Further researches in mice have also shown that the combination of drugs produces a better immune response, but researchers will still answer results from large human trials.
In the UK, Oxford Vaccine Group conducts a Com-CoV trial to study a combination of the first post-Pfizer or Covishield followed by a second dose of Moderna or Novavax. And it will be the largest study with at least 800 volunteers aged 50 and over, with a capacity of 1,000 more.
According to a letter from researchers published in The Lancet, preliminary results show that a combination of Covishield and Pfizer may produce a strong immune response – pain or swelling in the injection site, fever and headache – after the second dose of two doses of the same vaccine. But this response was not difficult or long-lasting and there were no other safety concerns. However, it is not clear whether the combination hits efficiency. More details are awaited later this month.
A lead researcher of the study, Dr. Matthew Snap, said, “If they can show that these mixed systems create an immune response similar to normal programs, and without a significant increase in vaccine mutations, this will allow more people to complete their Covid-19 vaccine studies more quickly.”
Can combining vaccines improve immune response?
According to some experts, a combination of antiretroviral drugs may better prepare the immune system to fight off a wide range of threats, including new ones.
As per a Spanish study, that involved more than 600 people, 400-odd found the first dose of Covishield followed by Pfizer shooting eight weeks later. They have generated much higher levels of antibodies than others, but it is still unclear whether the Covishield-Pfizer combination works better than the two doses of Pfizer.
According to Dr. V Ravi, who is a pathologist and also a member of the expert committee of the Department of Applied Technology said that personally, these vaccines have declared to be safe, and psychologically there should be no safety concerns. … By protecting the body, the goal is to go ahead with one dose and the other to increase it. The key question that needs to be answered is whether mixing will affect efficiency.
In some countries where there is a shortage of vaccines, the main purpose of mixing guns is to check whether it works or not. In India, for example, having many safe combinations of vaccines can help intensify vaccination efforts.
“It will provide flexibility in using any available vaccine. Remember, the regulator has given his approval of the same drug in two doses, so a bridge test may really be needed. Therefore the details – whether done in India or abroad – must be disclosed before the regulator here, ”said Dr. Ravi.
The UK, Germany, and France have already agreed to combine the drugs in ‘different situations’ and several trials, especially for adults, continue to better understand the outcomes. The US and UK have advised against mixing doses unless unavoidable.
In Europe, anxieties about safety around Covishield have urged Germany and France to recommend young people who have received Covishield first to receive a second dose of mRNA. Some countries await the outcome of the trial before formally supporting the mix.
According to some experts, mixing may be needed more time to check if the virus develops into a new species or if limited immunity from a two-drug source requires a booster in the future next year.

Post Your Comments