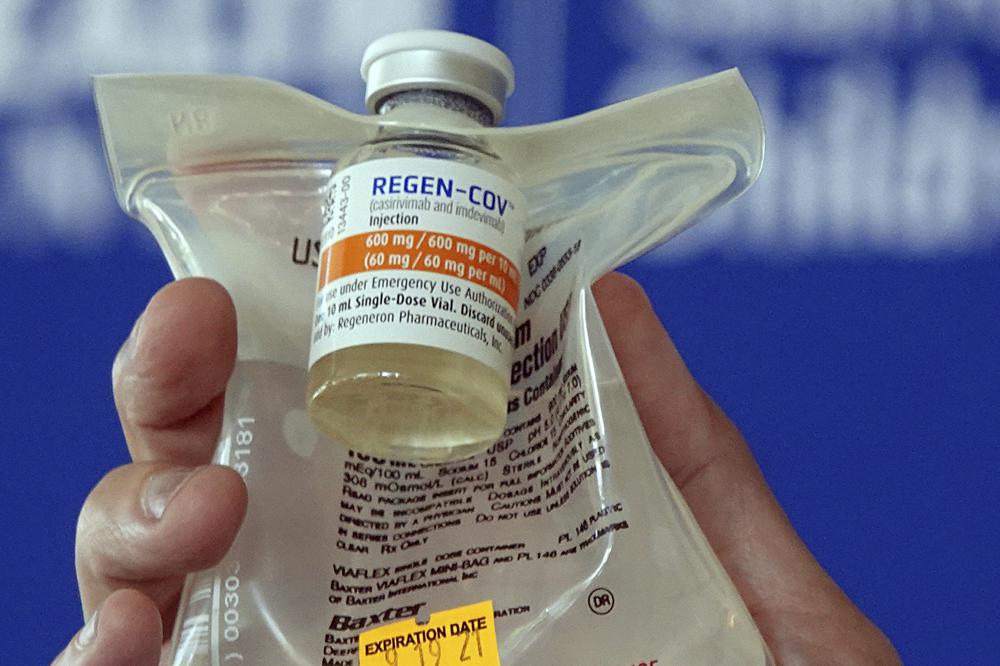
U.S. health regulators warned on Monday that 19 antibody medications from Regeneron and Eli Lilly should no longer be used because they don’t function against the omicron form, which causes nearly all infections in the United States.
The Food and Drug Administration announced that it was cancelling emergency authorisation for both medications, which were acquired by the federal government and given to millions of Americans who were infected with COVID-19. The FDA said it could reauthorize the medications’ use if they prove successful against subsequent versions.
As both drug-makers have said that infusion medicines are less able to target omicron due to its mutations, the regulatory action was expected. Nonetheless, some Republican governors may object to the federal move because they have continued to promote the medications despite the advise of health professionals.
In recent weeks, Omicron’s resistance to two of the most commonly used monoclonal antibody drugs has thrown the COVID-19 treatment plan into disarray.
Alternative medicines, such as two new antiviral medications from Pfizer and Merck, are available to treat early COVID-19 cases, although both are in short supply. GlaxoSmithKline’s antibody medicine, which is still effective, is likewise in short supply.
The medications are cloned versions of virus-blocking antibodies created in a lab. They’re designed to prevent serious illness and death by delivering concentrations of one or two antibodies early in an infection. After testing positive for the coronavirus in 2020, then-President Donald Trump was given Regeneron’s antibody combo.
As omicron raced across the country to become the dominant form, the US government temporarily halted distribution of the two medications in late December. However, after objections from Republican governors, including Florida’s Ron DeSantis, who claimed that the medications were still helping certain omicron patients, officials restarted delivery.
While rejecting vaccine requirements and other public health initiatives, DeSantis has extensively promoted antibody medicines as a signature aspect of his administration’s COVID-19 response, setting up infusion facilities and praising them at press conferences. Greg Abbott, the governor of Texas, has also opened state-sponsored infusion clinics.
The medications are not a replacement for immunisation and are typically reserved for the most vulnerable, such as the elderly, transplant recipients, and those suffering from heart disease or diabetes.
The US government has supplied enough dosages of the two antibodies to treat approximately 300,000 individuals since early January.
Both Regeneron and Eli Lilly had previously stated that they were working on new anti-omicron antibodies.
The decision comes just days after regulators expanded the use of remdesivir, the first COVID-19 medication, to include more patients.
The FDA extended the antiviral’s approval to adults and children with early COVID-19 who are at high risk of being admitted to the hospital on Friday. Remdesivir was previously only available to people in hospitals.
The injected medicine had already been suggested by a powerful group of federal specialists to try to avoid hospitalisation. Due to their lower effectiveness against omicron, the National Institutes of Health panel advises against continuing to utilise Eli Lilly and Regeneron’s antibody medicines.
Despite this, many hospitals will find difficulties in implementing remdesivir treatments. When used for non-hospitalized patients, the medication requires three consecutive IV infusions over three days. Many over-capacity hospitals with personnel shortages will be unable to complete this time-consuming process.
The FDA announced its conclusion based on a 560-patient study that found remdesivir reduced hospitalizations by nearly 90 percent when taken within seven days of onset of symptoms.
Although the trial predates the omicron variety, remdesivir, like other antivirals, is predicted to maintain its effectiveness against the most recent variant.

Post Your Comments