The phase II clinical trial of the Oxford COVID vaccine started in India on Wednesday at a Pune hospital.Vital health parameters of the two volunteers, that have been administered are normal.
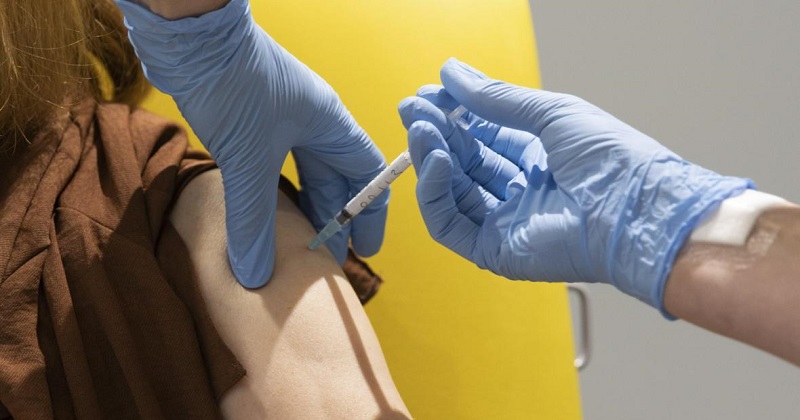
One of the volunteers was a 48-year-old gynecologist at a private hospital in Pune who had also volunteered 10 years ago for clinical trials for a vaccine against the H1N1 virus. The other is a 32-year-old doctoral candidate in statistics who works for a private company in the city and also is the first volunteer to be injected with the candidate that has been named Covishield in India.
After the vaccine was administered, both the volunteers were observed for 30 minutes and later allowed to go home, adding that the hospital’s medical team is conducting follow-ups with the volunteers.
A total of 1,600 volunteers will be part of the study at multiple trial sites. The Drug Controller General of India (DCGI), the national drug regulator, had on August 3 allowed Pune-based Serum Institute of India, the world’s largest manufacturer of vaccines, to conduct phase 2/3 human clinical trials of the vaccine candidate in India. Oxford University researchers announced last month that the candidate had triggered a dual immune response in phase 1 and phase 2 human clinical trials against the coronavirus that causes Covid-19.

Post Your Comments